Photobiomodulation, Biostimulation, Laser Therapy - Background and Basics
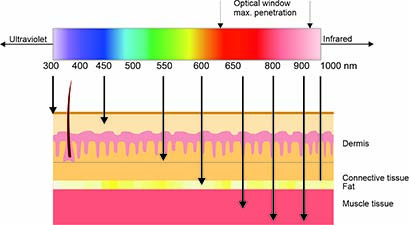
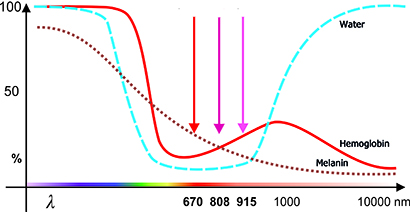
Photobiomodulation therapy (PBMT), also known as biostimulation or low-level laser therapy (LLLT, cl. 3B laser devices) and high-level laser therapy (HLLT, cl. 4 laser devices), is a non-invasive treatment method. It utilizes light sources such as lasers or LEDs in the visible red (600–700 nm) and near-infrared (700–1100 nm) spectrum. It should be noted that wavelengths above 950 nm have a shallow penetration depth and unnecessarily heat the tissue (due to absorption by water).
Unlike surgical lasers, PBMT does not destroy tissue but instead induces photophysical and photochemical reactions at the cellular level. The power output of LLLT laser is typically a maximum of 500 mW, while HLLT can reach several watts; typical applications operate at approximately 15 W in continuous wave mode.
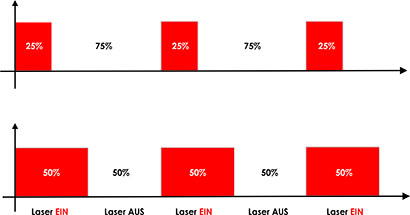
A special form of LLLT involves pulsed lasers as the LaserPen or Physiolaser, which can achieve peak power outputs of up to 100 W. The pulse width (duty cycle, DC) ranges from approximately 100 to 200 nanoseconds, depending on the pulse frequency.
A modern therapeutic laser should offer not only a suitable wavelength and sufficient power but also the possibility of modulation and frequency control. Ideally, therapeutically relevant frequencies are pre-programmed and easily accessible.
Three parameters are particularly crucial for the success of the therapy:
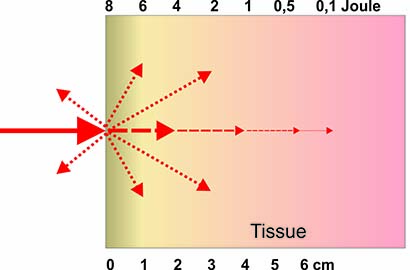
• The optimal laser wavelength, which largely determines the penetration depth of the light (optical window).
• The appropriate laser power, which influences cell activity and the energy increase.
• The correct laser modulation frequency, which enables targeted stimulation and control of the therapy at the informational level.
Biological Effects of Laser Therapy
The laser beam represents a gentle yet powerful therapeutic tool. Even a low-power laser of just 1 mW at a wavelength of 670 nm emits approximately
3×1015 photons per second, illustrating its significant biological potential. The skin penetration depth depends on various components as e.g. color and tissue type.
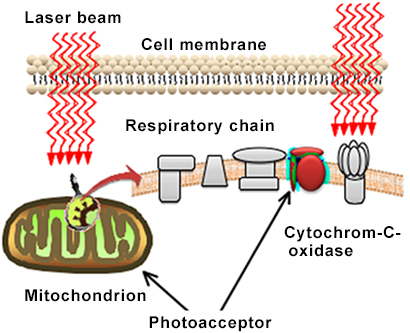
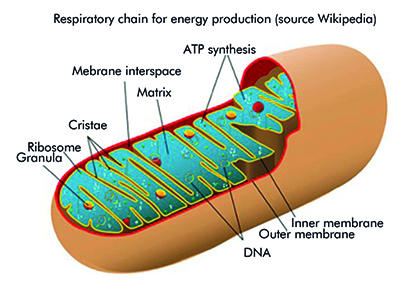
Appropriate laser application can induce a variety of responses in living organisms. The delivered laser energy is absorbed by tissues and cells, leading to enhanced cellular metabolism. This effect is primarily mediated through activation of the mitochondrial respiratory chain and a subsequent increase in ATP production.
According to Tina Karu and colleagues, cytochrome c oxidase is considered a key photoreceptor when cells are exposed to monochromatic red and near-infrared radiation. Several primary mechanisms of action have been proposed:
Alterations in the redox properties of respiratory chain components.
Photoexcitation of electron states, resulting in the generation of singlet oxygen and localized, transient heating of absorbing chromophores.
Increased production of superoxide anions, followed by elevated levels of hydrogen peroxide (H₂O₂) as a dismutation product.
Activation of a cascade of intracellular processes involving changes in cellular homeostasis parameters such as intracellular pH (pHi), calcium concentration [Ca2+]i , cyclic AMP (cAMP), redox potential (Eh), and ATP levels. These processes are interpreted as part of a photobiological signal transduction and amplification pathway (secondary mechanisms).
These mechanisms collectively contribute to the therapeutic effects observed in photobiomodulation.
References
Tina Karu, Institute of Laser and Information Technologies, Russian Academy of Sciences, Troitsk, Moscow Region, Russian Federation
Further relevant work can be found in the publications of:
Herbert Klima, Atomic Institute of the Austrian Universities, Vienna, Austria
Helmut Walter, Germany